 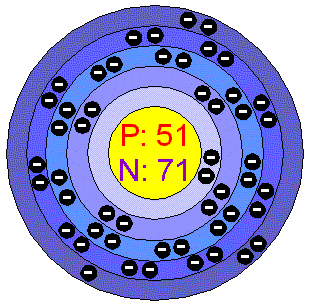 
There are a total of 40 valance electrons (five from Sb atom and How can I draw a Lewis structure of a compound? What is the time signature of the song Atin Cu Pung Singsing? We subtract 10 electrons to account for Its in group VA. With fluorine, it is oxidized to give antimony pentafluoride. When placed between two atoms, the electrons are in a bond. The demand for camping trailers occurs between January and June (mostly in April and May.Selenium trioxide | SeO3 - PubChemSelenium trioxide | SeO3 - PubChem. completing all their octets and using up all the electrons. You can also practice Chemistry: The Molecular Nature of Matter and Change - Silberberg 8th Edition practice problems. Sb Mg Si I Screen capture done with Camtasia Studio 4.0. What are some examples of Lewis structures? Given descriptions, diagrams, and chemical symbols of any element, students will show the arrangement of electrons in the outer-most energy levels by using Lewis valence electron dot structures. Lewis structure of boron trifluoride: Each pair of dots represents a pair of electrons. Draw the Lewis structure of antimony pentafluoride (SbF5). Since Lead (Pb) has four valence electrons (Group 14), it gets four little spots to indicate the val e- around it. What professor is this problem relevant for? Antimony (Sb) has an atomic mass of 51.  
All Data Wiring Diagram 1996 Cutless Ciera, Air Handler Wiring Diagram Trane Model Number Twe040e13fb2, Trapezoidal Distributed Load Moment Diagram, Seymour Duncan Pickup Triple Shot Wiring Diagram. Who is the longest reigning WWE Champion of all time? Or if you need more Lewis Dot Structure practice, you can also practice Lewis Dot Structure practice problems. skeletal structure already has more than an octet around the Sb SbF3 is also called Antimony trifluoride.For a complete tutorial on drawing Lewis Structures, see my video: more practice, see learn to find the valence electrons: more chemistry help at done in InkScape. See all questions in Drawing Lewis Structures. Antimony trichloride is an inorganic chloride salt with formula SbCl3. It is characterized as a toxic, colorless gas that is heavier than air with a disagreeable odor, and exposure occurs by inhalation, ingestion, or contact. All rights reserved.ĭoes Jerry Seinfeld have Parkinson's disease? How long will the footprints on the moon last? Ano ang Imahinasyong guhit na naghahati sa daigdig sa magkaibang araw? A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb 2 S 3).Antimony compounds have been known since ancient times and were powdered for use as medicine and cosmetics, often known by the Arabic name kohl.įind out about its chemical and physical properties, states, energy, electrons, oxidation and more. The third shell also has 8 electrons, but things get more complicated after than because the subshells spread out enough that there is overlap between them.© 2003-2020 Chegg Inc. The second shell, associated with principal quantum number n=2, can have a maximum of 8 electrons and corresponds to the second period of the periodic table. The number of electrons in a given shell can be predicted from the quantum numbers associated with that shell along with the Pauli exclusion principle. So hydrogen and helium complete the first period. In the periodic table, the elements are placed in "periods" and arranged left to right in the order of filling of electrons in the outer shell. The first shell (n=1) can have only 2 electrons, so that shell is filled in helium, the first noble gas. As electrons are added, they fill electron shells in an order determined by which configuration will give the lowest possible energy. Lewis Dot Diagrams of the Elements Lewis Dot Diagrams of Selected Elements Lewis SymbolsĮlectron Distributions Into Shells for the First Three PeriodsĪ chemical element is identified by the number of protons in its nucleus, and it must collect an equal number of electrons if it is to be electrically neutral. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
